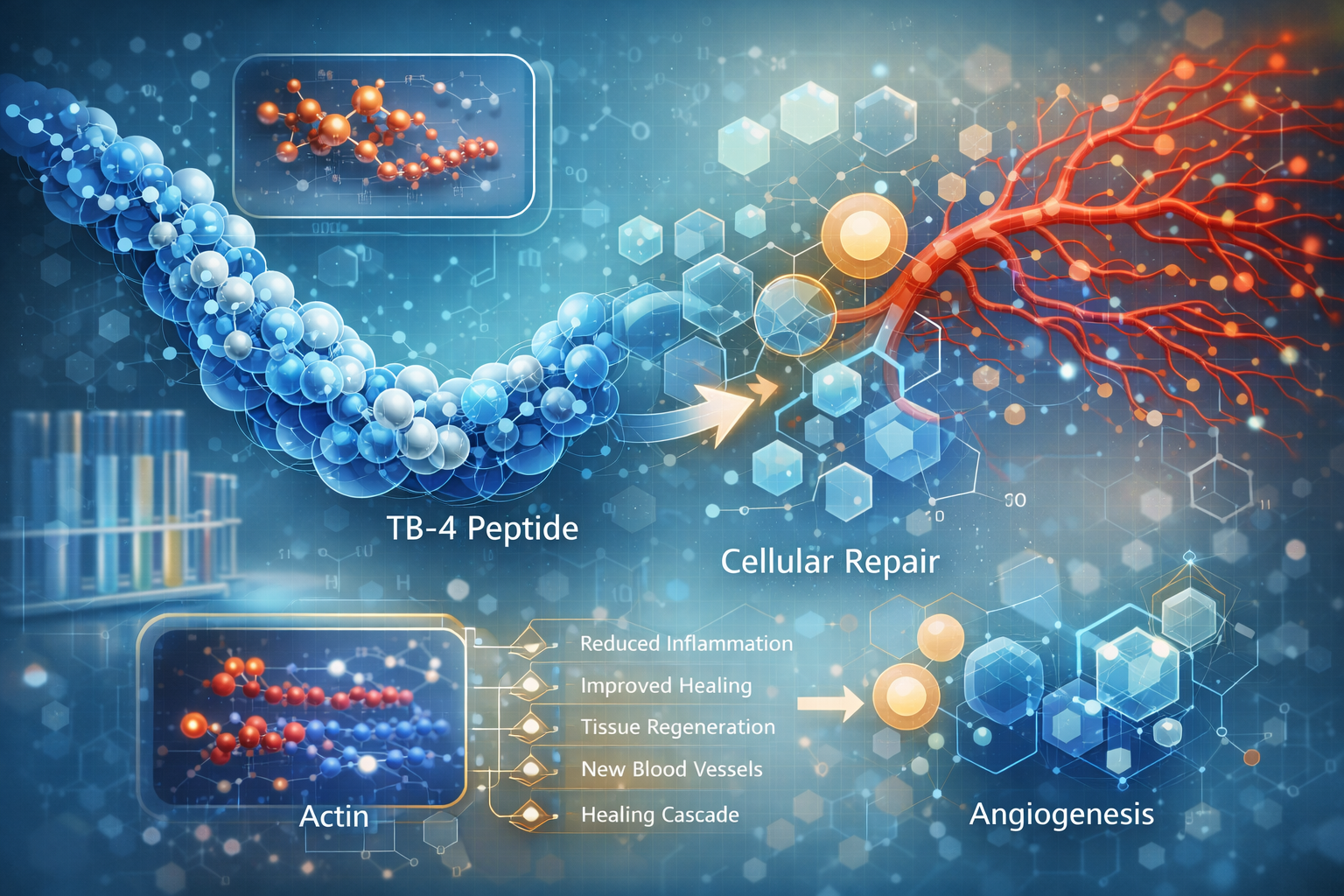
Thymosin Beta 4 represents one of the most extensively researched tissue repair peptides in 2026. This naturally occurring 43-amino acid peptide plays crucial roles in cellular migration, angiogenesis, and tissue regeneration, making Thymosin Beta 4 essential for researchers studying wound healing and regenerative processes. Understanding how Thymosin Beta 4 works at the molecular level provides insights into fundamental repair mechanisms applicable across multiple tissue types and injury models.
Molecular Structure and Origin of Thymosin Beta 4
Thymosin Beta 4 is a highly conserved peptide found in virtually all mammalian cells except red blood cells. It comprises 43 amino acids with the sequence critical for its actin-binding properties. The peptide is particularly concentrated in platelets, wound fluid, and other tissue repair sites.
In the body, Thymosin Beta 4 is released by platelets during injury, creating a concentration gradient that directs healing processes. This natural mechanism has made Thymosin Beta 4 a focal point for regenerative medicine research.
Primary Mechanisms of Action: Thymosin Beta 4
Actin Sequestration:
Thymosin Beta 4’s most fundamental mechanism involves binding to G-actin (globular actin) monomers. This prevents premature polymerization into F-actin (filamentous actin), maintaining a pool of available actin for controlled cytoskeletal remodeling. This Thymosin Beta 4 process is essential for:
- Cell migration and motility
- Cellular shape changes
- Wound contraction
- Tissue remodeling
Angiogenesis Promotion:
Thymosin Beta 4 powerfully stimulates new blood vessel formation through multiple pathways:
- Upregulates vascular endothelial growth factor (VEGF)
- Promotes endothelial cell migration
- Enhances endothelial tube formation
- Supports vessel stabilization and maturation
Research demonstrates Thymosin Beta 4 can increase vascular density by 30-40% in tissue repair models.
Anti-Inflammatory Properties:
Thymosin Beta 4 modulates inflammatory responses through:
- Downregulation of pro-inflammatory cytokines (TNF-α, IL-1β, IL-6)
- Upregulation of anti-inflammatory mediators
- Reduced neutrophil infiltration in acute inflammation
- Protection against excessive inflammatory tissue damage
Stem Cell Recruitment:
Thymosin Beta 4 attracts endogenous stem cells to injury sites:
- Enhances stem cell survival in hostile injury environments
- Promotes stem cell differentiation into needed cell types
- Increases stem cell migration velocity
- Supports stem cell integration into damaged tissue
Research Applications 2026: Thymosin Beta 4
Cardiovascular Research:
Extensive research explores Thymosin Beta 4 in cardiac injury models:
- Myocardial infarction studies show 20-30% reduction in scar size
- Enhanced cardiac function post-injury in animal models
- Promotion of epicardial progenitor cell activation
- Improved coronary vessel formation
Musculoskeletal Research:
Thymosin Beta 4 demonstrates significant effects in connective tissue studies:
- Accelerated tendon healing in injury models
- Improved muscle regeneration after strain injuries
- Enhanced ligament repair processes
- Reduced fibrosis and scar tissue formation
Research shows healing time reductions of 30-50% in standardized injury models using Thymosin Beta 4.
Dermal Wound Healing:
Skin repair studies document multiple beneficial Thymosin Beta 4 effects:
- Faster wound closure rates (40-60% improvement)
- Enhanced re-epithelialization
- Better organized collagen deposition
- Reduced scarring compared to controls
- Improved hair follicle regeneration
Neurological Research:
Emerging Thymosin Beta 4 applications in neural injury research:
- Neuroprotective effects in ischemic models
- Enhanced neuronal survival after injury
- Promotion of oligodendrocyte maturation
- Potential benefits in neurodegenerative research
Ophthalmological Studies:
Corneal and retinal Thymosin Beta 4 research applications:
- Accelerated corneal wound healing
- Dry eye syndrome research models
- Retinal injury protection studies
- Reduced inflammation in eye injury models
Dosing Protocols in Research: Thymosin Beta 4
Standard Loading Phase:
- Dosage: 2-5mg administered twice weekly
- Duration: 4-6 weeks
- Route: Subcutaneous or intramuscular injection
- Purpose: Establish systemic therapeutic levels
Maintenance Protocol:
- Dosage: 2-5mg once weekly
- Duration: Variable based on research objectives
- Route: Subcutaneous preferred for convenience
- Purpose: Maintain elevated systemic levels
Acute Injury Protocol:
- Dosage: 5-10mg for first 7-10 days
- Frequency: Daily or every other day
- Route: Near-site injection when possible
- Duration: 2-4 weeks intensive, then standard maintenance
Research Findings on Optimal Thymosin Beta 4 Dosing:
Studies indicate Thymosin Beta 4 exhibits dose-dependent effects up to approximately 5-7.5mg per injection, with diminishing returns at higher doses. Frequency appears more important than individual dose magnitude for sustained tissue repair effects.
Pharmacokinetics: Thymosin Beta 4
Absorption and Distribution:
- Rapid absorption following subcutaneous injection
- Wide tissue distribution due to small molecular size
- Concentrates in injury sites due to inflammation gradients
- Crosses blood-brain barrier in limited amounts
Half-Life:
- Plasma half-life: Approximately 2-3 hours
- Tissue half-life: Significantly longer (days)
- Biological activity: Extends beyond plasma clearance
- Effects persist 24-72 hours post-administration
TB-4 vs TB-500: Critical Distinction in Thymosin Beta 4 Research
The research community often uses these terms interchangeably, but important differences exist:
Thymosin Beta-4 (TB-4):
- Full 43-amino acid natural sequence
- Complete biological activity
- More expensive to synthesize
- Identical to endogenous peptide
TB-500:
- Synthetic fragment (typically 17-23 amino acids)
- Contains the active region of TB-4
- More economical to produce
- Demonstrates similar primary effects
- May lack some secondary activities
Most Thymosin Beta 4 research uses TB-500 due to cost-effectiveness while maintaining primary mechanisms of interest.
Reconstitution and Storage: Thymosin Beta 4
Lyophilized Powder:
- Storage temperature: -20°C
- Stability: 24-36 months when properly stored
- Protect from moisture and light
- Sealed in inert atmosphere
Reconstituted Solution:
- Solvent: Bacteriostatic water or sterile water
- Concentration: Typically 2-5mg per mL
- Storage: 2-8°C refrigerated
- Stability: 30-60 days (bacteriostatic water extends stability)
- Do not freeze once reconstituted
Safety and Side Effects: Thymosin Beta 4
Thymosin Beta 4 research demonstrates excellent safety profiles:
Minimal side effects observed:
- Occasional injection site reactions
- Mild headache (less than 5% of research subjects)
- Transient lethargy (rare)
- No significant organ toxicity in long-term studies
No observed adverse effects:
- No hormonal disruption
- No immune system suppression
- No tumor promotion in standard research models
- No significant drug interactions documented
Contraindications in research models:
- Active malignancy (theoretical concern due to angiogenic effects)
- Uncontrolled infection (could potentially enhance pathogen growth)
Combination Protocols: Thymosin Beta 4
Thymosin Beta 4 is frequently combined with other peptides for synergistic research:
TB-4 + BPC-157:
- Complementary tissue repair mechanisms
- BPC-157 focuses on GI and tendon healing
- TB-4 provides broader angiogenic effects
- Combined protocols show enhanced overall healing
TB-4 + GHK-Cu:
- Both promote tissue remodeling
- Copper peptide enhances collagen formation
- TB-4 provides vascular support
- Excellent for skin and connective tissue research
TB-4 + Growth Hormone Peptides:
- GH provides anabolic environment
- TB-4 directs tissue repair processes
- Synergistic for recovery research
- Combined effects on collagen synthesis
Injection Technique: Thymosin Beta 4
Subcutaneous Administration:
- Common sites: Abdomen, thighs, upper arms
- Needle: 28-31 gauge, 0.5 inch
- Technique: 45-90 degree angle
- Advantages: Convenient, painless, consistent absorption
Intramuscular Administration:
- Sites: Deltoid, gluteus, vastus lateralis
- Needle: 23-25 gauge, 1-1.5 inch
- Technique: 90-degree angle, deep injection
- Advantages: Potentially faster absorption for acute research
Near-Site Injection:
- For localized injury research
- Inject within 2-3cm of injury site when possible
- May enhance local concentration
- Evidence for superiority over systemic is mixed
Clinical Trial Progress 2026: Thymosin Beta 4
Several Thymosin Beta 4 clinical trials are ongoing or completed:
Phase III Dry Eye Trial:
- RGN-259 (TB-4 derivative) for dry eye syndrome
- Showing promising efficacy results
- Well-tolerated with minimal side effects
- Potential FDA approval pathway
Cardiovascular Trials:
- Post-MI (myocardial infarction) recovery studies
- Early-phase trials showing safety and preliminary efficacy
- Ongoing research in heart failure models
Dermatological Applications:
- Venous stasis ulcer healing trials
- Diabetic wound research programs
- Epidermolysis bullosa treatment studies
Comparative Research: Thymosin Beta 4 vs Other Repair Peptides
TB-4 vs BPC-157:
- TB-4: Broader angiogenic effects, systemic distribution
- BPC-157: More focused on GI protection and tendon healing
- TB-4: Better vascular research applications
- BPC-157: Superior gastric protection
TB-4 vs GHK-Cu:
- TB-4: Stronger angiogenesis and cell migration
- GHK-Cu: Superior collagen remodeling and antioxidant effects
- TB-4: Better acute injury response
- GHK-Cu: Better for skin and anti-aging research
Performance Research Applications: Thymosin Beta 4
Athletic and recovery research explores Thymosin Beta 4 for:
- Accelerated post-exercise recovery
- Reduced delayed onset muscle soreness (DOMS)
- Enhanced connective tissue resilience
- Faster return to training after injury
Research shows recovery time improvements of 25-40% in controlled injury-recovery protocols using Thymosin Beta 4.
WADA Status:
Thymosin Beta 4 is prohibited by the World Anti-Doping Agency under the S0 category (non-approved substances). Detection methods exist, though the peptide’s short plasma half-life creates detection challenges.
Cost Considerations: Thymosin Beta 4
Research budget for Thymosin Beta 4 protocols:
Loading Phase (6 weeks, 2x weekly at 5mg):
- Total peptide: ~60mg
- Approximate cost: $300-500
Maintenance (12 weeks, 1x weekly at 5mg):
- Total peptide: ~60mg
- Approximate cost: $300-500
6-month Thymosin Beta 4 research protocol: $600-1000
TB-500 (synthetic fragment) typically costs 30-50% less while maintaining primary research utility.
Quality and Purity Considerations: Thymosin Beta 4
Research-grade Thymosin Beta 4 should meet:
- Minimum purity: 98% (HPLC verified)
- Correct amino acid sequence confirmation
- Endotoxin testing completed
- Third-party testing certificates available
- Proper lyophilization and packaging
USA-manufactured Thymosin Beta 4 peptides from verified suppliers ensure consistent research quality.
Future Research Directions: Thymosin Beta 4
Emerging Thymosin Beta 4 research areas include:
- Combination with stem cell therapies
- Nanoparticle delivery systems for targeted release
- Modified TB-4 analogs with enhanced stability
- Oral bioavailability formulations
- Tissue-specific targeting mechanisms
Frequently Asked Questions: Thymosin Beta 4
How long does Thymosin Beta 4 take to show effects?
Research models typically demonstrate measurable effects within 7-14 days of initiating protocols. Maximum effects are generally observed at 4-6 weeks with consistent administration.
Can Thymosin Beta 4 be used for chronic injuries?
Research shows Thymosin Beta 4 effectiveness in both acute and chronic injury models, though acute injuries typically respond more dramatically. Chronic injury research may require longer protocols.
Is Thymosin Beta 4 safe for long-term research?
Studies extending 6-12 months show maintained safety profiles with no significant adverse effects. Long-term Thymosin Beta 4 research appears well-tolerated in animal models.
Conclusion
Thymosin Beta 4 stands as a cornerstone peptide for tissue repair and regenerative research in 2026. Its multi-faceted mechanisms—actin regulation, angiogenesis, anti-inflammatory effects, and stem cell recruitment—make Thymosin Beta 4 uniquely valuable for investigating healing processes across multiple tissue types.
The peptide’s excellent safety profile, combined with substantial research demonstrating efficacy in numerous injury models, positions Thymosin Beta 4 as an essential tool for researchers studying tissue repair, cardiovascular recovery, wound healing, and regenerative medicine.
For research applications, Thymosin Beta 4 should be sourced from verified USA manufacturers maintaining strict purity standards and proper handling protocols throughout the supply chain.
DISCLAIMER: FOR RESEARCH USE ONLY – NOT FOR HUMAN CONSUMPTION
This content is for educational and research purposes only. These products are sold strictly for in-vitro research and laboratory experimentation only. All information provided is for informational purposes and is not intended for diagnostic or medical use. Peptides described are not for human consumption. Vivo Peptides does not encourage or promote the use of any of these products in a manner contrary to the law.